| Table of Contents |  |
|
Case Report
| ||||||
| Disseminated gonococcal infection in 21st century: A case report | ||||||
| Sumir Kumar1, Bharat Bhushan Mahajan2, Sandeep Kaur3 | ||||||
|
1MD, Assistant Professor, Department of Dermatology, Venereology & Leprology, GGS Medical College & Hospital, Faridkot, Punjab, India.
2MD, Professor & Head, Department of Dermatology, Venereology & Leprology, GGS Medical College & Hospital, Faridkot, Punjab, India. 3MBBS, Postgraduate Resident, Department of Dermatology, Venereology & Leprology, GGS Medical College & Hospital, Faridkot, Punjab, India. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Kumar S, Mahajan BB, Kaur S. Disseminated gonococcal infection in 21st century: A case report. Edorium J Infect Dis 2014;1:1–4. |
|
Abstract
|
|
Introduction:
Disseminated gonococcal infection (DGI) is an uncommon complication associated with Neisseria gonorrhoeae. But due to early diagnosis and use of newer drugs in gonorrhea, complications like DGI have become a rarity. It is more common in women than men. We herein report a case of this entity occurring in otherwise healthy male who presented with septic shock.
Case Report: An 18-year-old healthy male presented with urethral discharge, conjunctivitis, oral ulcerations, and tenosynovitis along with septic shock. History of unprotected sexual contact was present two weeks prior to the onset of above mentioned complaints. A clinical diagnosis of DGI was kept which was confirmed by the presence of gram negative diplococci in urethral and conjunctival discharge smears as well as isolation of gonococci on selective culture media. He responded well to injectable ceftriaxone therapy. Conclusion: The presentation of DGI as septic shock in an immunocompetent male in the absence of predisposing factors is highlighted in this case report. Early diagnosis and treatment is the key to prevent devastating complications like meningitis, endocarditis, perihepatitis, etc. Moreover, infrequent occurrence of DGI calls for the need of introduction of diagnostic criteria in order to facilitate its prompt identification and treatment. | |
|
Keywords:
Disseminated gonococcal infection (DGI), Gonorrhea, Neisseria gonorrhoeae, Shock
| |
|
Introduction
| ||||||
|
Disseminated gonococcal infection (DGI) is caused by hematogenous spread of Neisseria gonorrhoeae which may be lead to symptoms and signs, such as tenosynovitis, dermatitis, and arthritis (in order of decreasing frequency). Approximately, 62 million new cases occur annually worldwide [1]. In India also, its prevalence ranges from 3–19% among sexually transmitted infection (STI) clinic attendees. However, DGI occurs in 0.5–3% of patients infected with Neisseria gonorrhoeae. But due to early diagnosis, and use of newer drugs in the treatment of gonorrhea, complications like DGI have become a rarity. We are reporting a case of a young immunocompetent male with DGI and septic shock for the rarity of this condition. | ||||||
|
Case Report
| ||||||
|
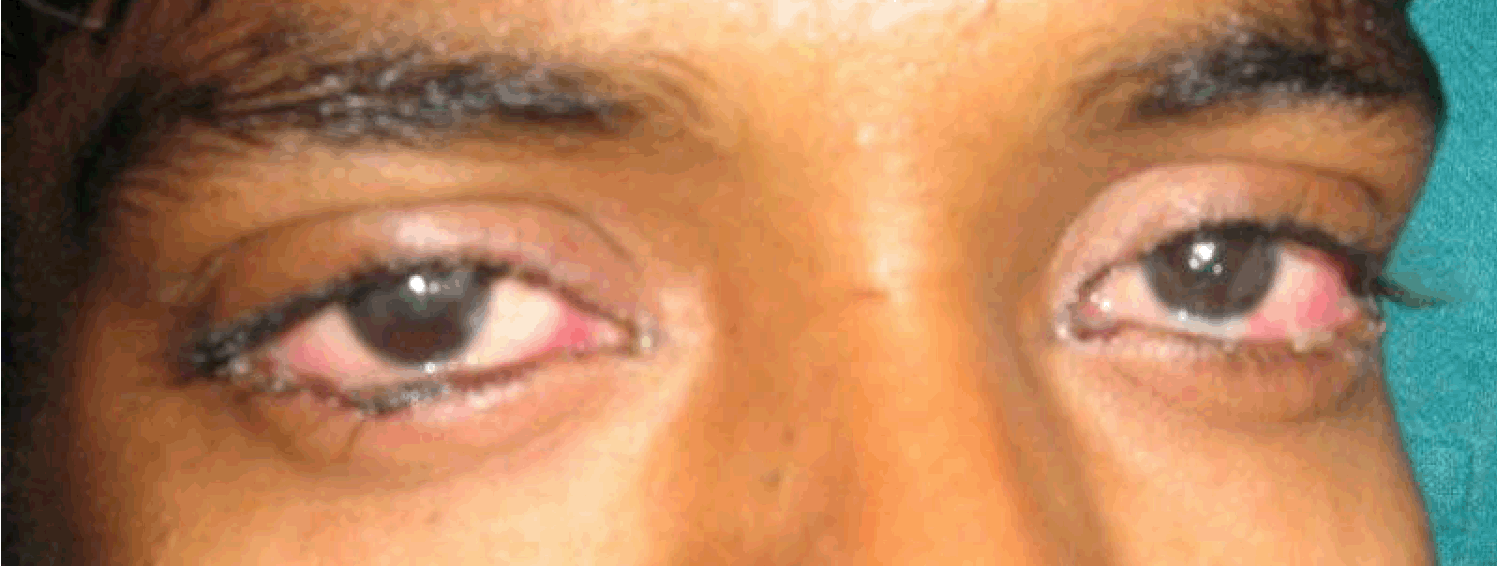
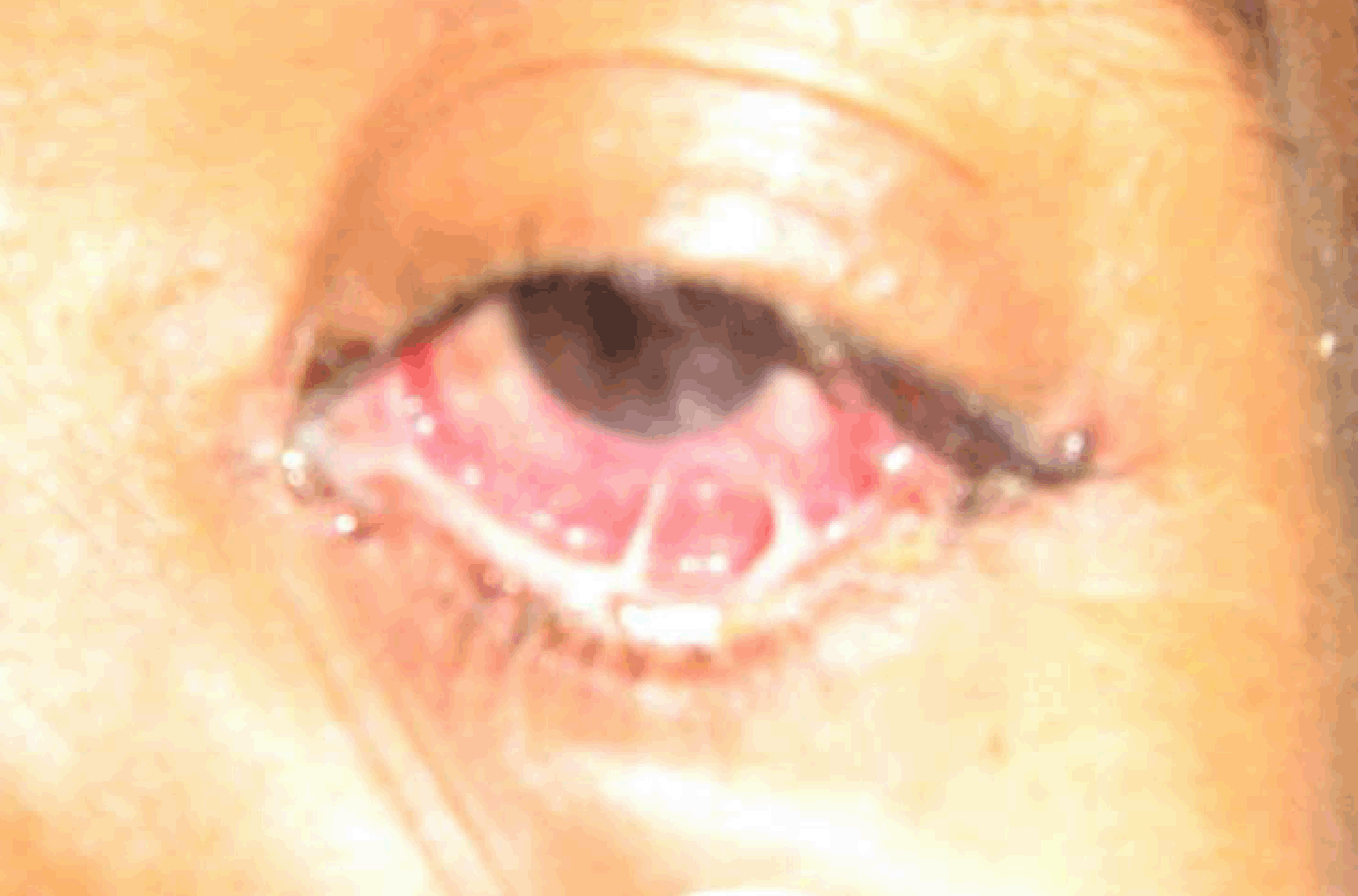
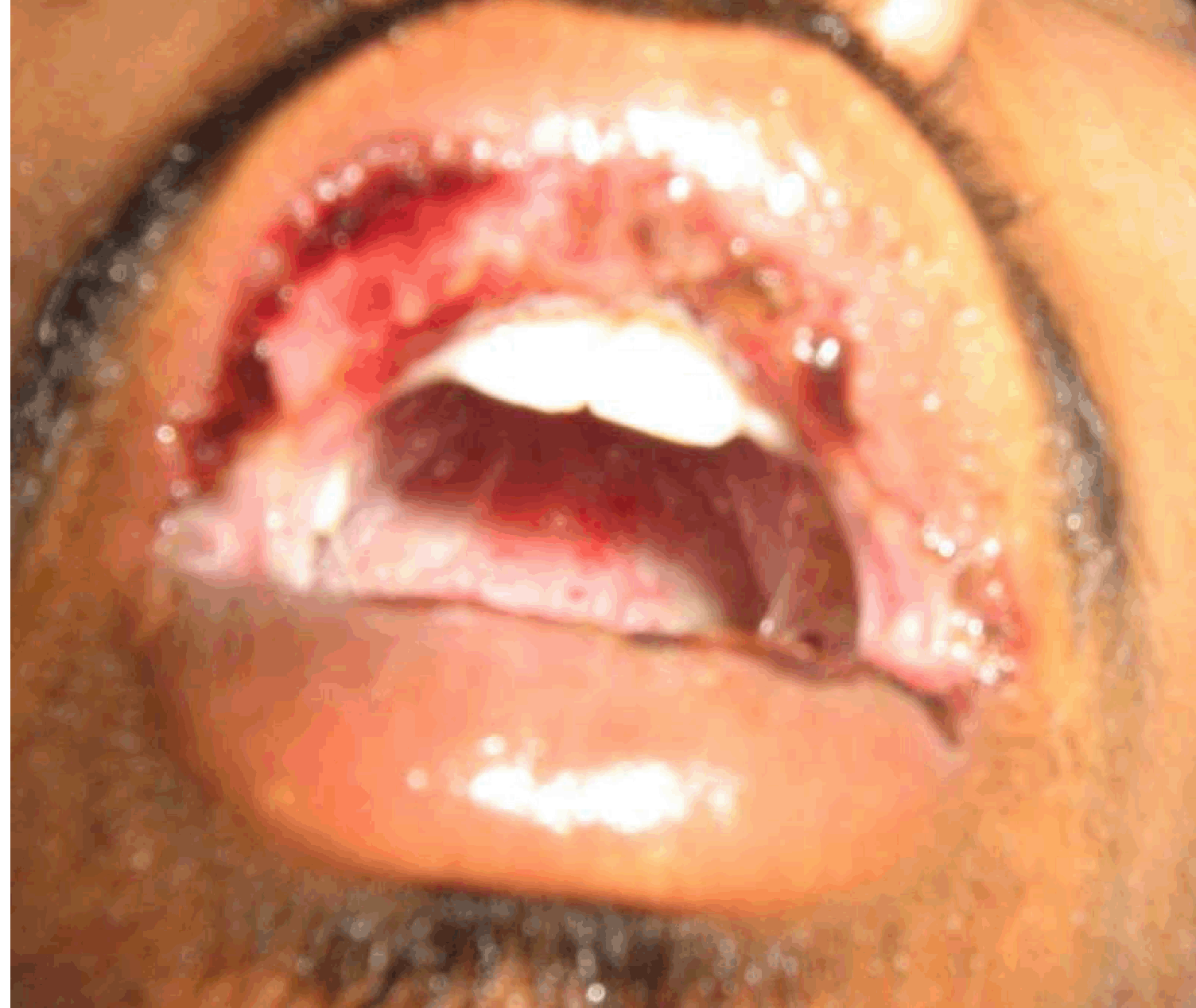
An 18-year-old male presented with complaints of greenish yellow discharge from eyes, multiple painful ulcers along with blood stained discharge in mouth, dysuria and mucopurulent urethral discharge for one week. There was history of high grade fever, malaise, myalgias and arthralgia. The patient was unmarried and welder by occupation. He had regular unprotected sexual contact and the last contact was about two weeks ago. On examination, the patient was febrile and hypotensive with blood pressure of 80/60 mmHg. Bilateral preauricular, cervical as well as inguinal lymph nodes were enlarged and tender. Systemic examination was found to be normal. There was profuse, thick mucopurulent discharge from both eyes with congested conjunctiva (Figure 1) and (Figure 2). Thin translucent membrane was present over bilateral bulbar conjunctiva. Multiple ulcers with blood-tinged serous discharge and thick yellowish slough were seen on tongue, labial, buccal, palatal mucosae and posterior pharyngeal wall (Figure 3). Nasal mucosa was crusted. There was profuse, thick mucopurulent urethral discharge with secondary phimosis (Figure 4). There was tenosynovitis on dorsal aspect of wrist and ankles. Hemogram showed mild leukocytosis but other routine investigations were within normal limits. Urine was full of pus cells and RBC's with mild albuminuria. Gram stain of both ocular and urethral discharge smears showed intracellular gram-negative kidney-shaped diplococci. Pus culture on chocolate agar media also revealed the presence of gonococci. Blood and urine culture came out to be negative for gonococci. Results of serologic analysis for systemic lupus erythematosus (SLE), human immunodeficiency virus, hepatitis and syphilis were negative. Chest X-ray, ECG and ultrasound of abdomen were normal. Patient was taking multiple unknown oral medications from a private practitioner but was not relieved. After diagnosis, patient was put on intravenous ceftriaxone 1 g twice a day along with doxycycline to treat chlamydia which is commonly associated with gonococcal infection and diagnostic facilities to rule out chlamydial infection were not available at our hospital. The patient started responding after two days. Repeat smears from ocular, oral and genital discharge were negative after seven days of starting the treatment with significant clinical improvement. | ||||||
| ||||||
| ||||||
| ||||||
| ||||||
|
Discussion
| ||||||
|
Though gonorrhea is one of the common sexually transmitted infections in India, it is showing a declining trend in the last decade [2]. Similarly, cases of gonococcemia are also becoming a rarity. The probability of dissemination of a localized gonococcal infection depends upon specific host, microbial, and possibly immune factors.
Patients with disseminated gonococcal infection (DGI) typically present with one of two syndromes [3]:
Disseminated gonococcal infection is one of the rare and dreaded complications of gonorrhea which may further lead to permanent joint damage, endocarditis, myocarditis, pericarditis, hepatitis, meningitis or epidural abscess. It can also rarely be associated with septic shock leading to multiple organ failure and death [6]. Our patient initially went into septic shock but was relieved after proper management. Later on, phimosis occurred for which surgical consultation was taken. Gonorrhea is usually diagnosed by the clinical presentation, identification of the organism on gram stain and isolation on culture media like chocolate agar media, modified Thayer-Martin media, Chacko-Nair media, Martin-Lewis media or New York City media. Newer techniques include nucleic acid amplification test (NAAT), polymerase chain reaction (PCR), transcription mediated amplification and standard displacement amplification on endocervical, urethral and urine specimens. In patients with purulent arthritis, the best diagnostic method is synovial fluid analysis. However, synovial fluid cultures yield negative results in patients with tenosynovitis, dermatitis, and polyarthralgia form of DGI. The diagnosis of gonococcemia in our case was made on the basis of the characteristic clinical picture and was confirmed by gram's staining of ocular as well as urethral discharge smear and growth on culture media. Third generation cephalosporins like ceftriaxone are recommended as the initial empirical therapy in gonococcal infections due to emergence of resistance to penicillins and fluoroquinolones [7]. Our patient also responded to intravenous ceftriaxone in about one week. | ||||||
|
Conclusion
| ||||||
|
The presentation of disseminated gonococcal infection (DGI) as septic shock in an immunocompetent male in the absence of predisposing factors is highlighted in this case report. Early diagnosis and treatment is the key to prevent devastating complications like meningitis, endocarditis, and perihepatitis. Moreover, infrequent occurrence of DGI calls for the need of introduction of diagnostic criteria in order to facilitate its prompt identification and treatment. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Sumir Kumar – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Bharat Bhushan Mahajan – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published Sandeep Kaur – Substantial contributions to conception and design, Acquisition of data, Analysis and interpretation of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2014 Sumir Kumar et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|